Dec 20, 2018 - Annex A – Nominal cross-references to ISO 9001:2000.://www.ukas.com/download/generaldocuments/UKAS-January-2018-training-v9.pdf. ISO/IEC 17025 was first issued in 1999 by the International Organization for. The 6th ISO/CASCO WG 44 meeting was held on July 10-12, 2017 in ISO Central.
Hello People,i agree with the view that it is advisable to have a Quality Manual in place as it is the basic document for introduction of the lab. Secondly would also suggest that the Procedure manual (prepared in lieu of 2005 standard requirements) be kept intact or upgraded. This will ensure that the process is cascaded to all levels.in the recent interactions that i have had with a few auditors, it was evident that all audits will now be objective. Irrespective whether you have a documented procedure or not, it has to be verified.now i wanted to enquire whether there are any templates available for preparing the Quality manual as per 2017 requirements. Hello People,i agree with the view that it is advisable to have a Quality Manual in place as it is the basic document for introduction of the lab.
Secondly would also suggest that the Procedure manual (prepared in lieu of 2005 standard requirements) be kept intact or upgraded. This will ensure that the process is cascaded to all levels.in the recent interactions that i have had with a few auditors, it was evident that all audits will now be objective. Irrespective whether you have a documented procedure or not, it has to be verified.now i wanted to enquire whether there are any templates available for preparing the Quality manual as per 2017 requirements. I am sorry for the delay in this response.Do you have a copy of the standard? Indicates no requirement for a Quality Manual exists in ISO/IEC.Why have a manual if none is required for accreditation? You can document procedures, scope, policy and anything else your lab and the standard requires. You could have a booklet-sized central source in which these documents are gathered.
I have also seen them as tabs in a spreadsheet document.I have seen a table of contents essentially used as a Manual. If you use a process map, the procedure references could be listed there. I have also seen procedures referenced as part of actions to address risks, in documentation where risks are listed. There are many practical methods to replace a Manual that frankly was historically used by very few people.All of that said, if you must have a Manual, there are.
I agree with Andy that their usefulness is limited, hence my points above.
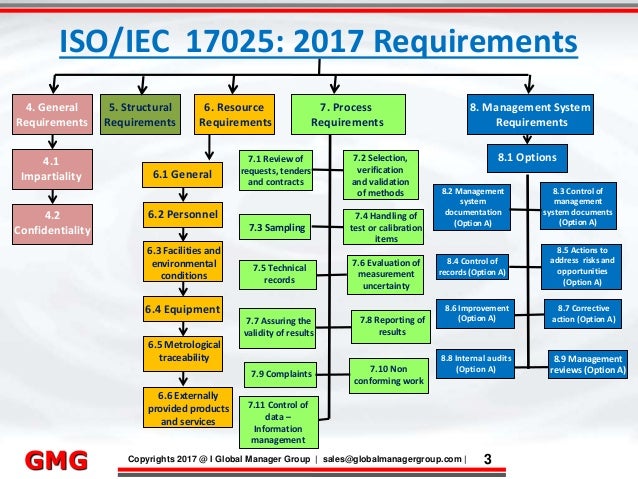
ISO/IEC 17025 General requirements for the competence of testing and calibration laboratories is the main used by testing and calibration laboratories. In most countries, ISO/IEC 17025 is the standard for which most labs must hold accreditation in order to be deemed technically competent. In many cases, suppliers and regulatory authorities will not accept test or calibration results from a lab that is not accredited. Originally known as ISO/IEC Guide 25, ISO/IEC 17025 was initially issued by the in 1999. There are many commonalities with the standard, but ISO/IEC 17025 is more specific in requirements for competence and applies directly to those organizations that produce testing and calibration results and is based on somewhat more technical principles. Laboratories use ISO/IEC 17025 to implement a quality system aimed at improving their ability to consistently produce valid results. It is also the basis for accreditation from an accreditation body.There have been three releases; in 1999, 2005 and 2017.
The most significant changes between the 1999 and 2005 release were a greater emphasis on the responsibilities of senior management, explicit requirements for continual improvement of the management system itself, and communication with the customer. It also aligned more closely with the 2000 version of ISO 9001The 2005 version of the standard comprises five elements; Normative References, Terms and Definitions, Management Requirements, and Technical Requirements. Management requirements are primarily related to the operation and effectiveness of the within the laboratory. Technical requirements include factors that determine the correctness and reliability of the tests and calibrations performed in the laboratory.The version of ISO/IEC 17025 has modified this structure to be Scope, Normative References, Terms and Definitions, General Requirements, Structural Requirements, Resource Requirements, Process Requirements, and Management System Requirements. General Requirements and Structural Requirements are related to the organization of the laboratory itself. Structure Requirements cite those issues related to the people, plant, and other organizations used by the laboratory to produce its technically valid results.
Process Requirements are the heart of this version of the standard in describing the activities to ensure that results are based on accepted science and aimed at technical validity. Management System Requirements are those steps taken by the organization to give itself tools to support the work of its people in the production of technically valid results. Contents.Predecessors Some national systems (e.g. M10 in the UK) were the forerunners of ISO/IEC but could sometimes be exceedingly prescriptive. ISO/IEC 17025 allows laboratories to carry out procedures in their own ways, but an ( assessor) may require the laboratory to justify using a particular method.In common with other ISO quality standards, ISO/IEC 17025 requires continual improvement. Additionally, the laboratory will be expected to keep abreast of scientific and technological advances in relevant areas.In common with other accreditation standards of the ISO 17000 series (and unlike most ISO standards for management systems), third party auditing ( assessment) of the laboratory is normally carried out by the national organization responsible for.
Laboratories are therefore accredited under ISO/IEC 17025, rather than certified or registered (c.f. ISO 9000 series).In short, accreditation differs from certification by adding the concept of a third party (Accreditation Body (AB)) attesting to technical competence within a laboratory in addition to its adherence and operation under a documented quality system, specific to a Scope of Accreditation.Accreditation bodies In order for accreditation bodies to recognize each other's accreditations, the (ILAC) worked to establish methods of evaluating accreditation bodies against another ISO/CASCO standard (ISO/IEC Guide 58 - which became ISO/IEC 17011). Around the world, geo-political regions such as the, and Asia-Pacific, the Americas and others, established regional cooperations to manage the work needed for such mutual recognition. These regional bodies (all working within the ILAC umbrella) include European Accreditation Cooperation (EA), the Asia Pacific Laboratory Accreditation Cooperation (APLAC), Southern Africa Accreditation Cooperation (SADCA) and the Inter-American Accreditation Cooperation (IAAC).The first laboratory accreditation bodies to be established were (NATA) in Australia (1947) and in New Zealand (1973).
Most other bodies are based on the NATA/TELARC model include and British Cert.in the UK, in Finland and in Denmark to name a few.In the U.S. There are several, multidisciplinary accreditation bodies that serve the laboratory community.
These bodies accredit testing and calibration labs, reference material producers, PT providers, product certifiers, inspection bodies, forensic institutions and others to a multitude of standards and programs. These ILAC MRA signatory accreditation bodies carry identical acceptance across the globe. It does not matter which AB is utilized for accreditation. The MRA arrangement was designed with equal weight across all economies. Canadian Association for Laboratory Accreditation (CALA). Retrieved 27 March 2018.
Honsa, Julie D.; Deborah A. McIntyre (2003). Journal of AOAC International. 86 (5): 1038–1044. Retrieved 28 February 2012.
SAI Global Limited ABN. Retrieved 28 February 2012.
Retrieved 2017-04-28. (PDF).
American Association for Laboratory Accreditation. Retrieved 28 February 2012. Retrieved 29 May 2015. Retrieved 29 May 2015. American Society of Crime Laboratory Directors-Laboratory Accreditation Board. Retrieved 12 July 2012. Retrieved 29 May 2015.External links.
